
Organic Phosphinate Compounds Are Ineffective in Treating Phosphorous Deficiency in Animals
As a veterinarian it is our professional responsibility to ascertain if a drug you are using in the practice is really effective, as the farmer is going to spend from his hard-earned money. Relying only on companies claims as gospel truth is not a correct approach. It is essential to seek hard data on the bioavailability from the point of ingestion to the site of action. It is also important to get satisfied with the mode of action of the drug. Sadly, there are many formulations marketed by even renowned companies that are known to be unavailable metabolically with no clue on mode of action and convincing double-blind placebo control trials data. The principle many companies follow is, ’if it sells, why worry’.
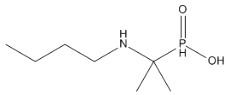
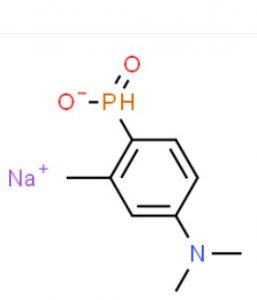 In this blog, I would like to discuss about two molecules many gullible veterinarians have been using as phosphorous tonic in their practice because of the marketing claims. These are: 4-dimethylamino-2-methyl-phenylphosphinic acid [Toldimfos], (available in the market as Tonophsphan, Injectiphos, Tonoricin, Tonogil, Lyphos, phosfovet, Rumiphos, T-Phos, Tenophos, etc.) and 1-butylamino-1-methyl-ethyl -phosphonic acid [Butaphosphan] (available under trade names, Cynophan, Catosal, etc.). From the list it will be clear that there are leading brands by multinational and clones marketed by local companies. The indicated claims in their brochure and literature are: phosphorous deficiency, hyophsophatemia, rickets, Ca-P imbalance, debility, anoestrus, and several other non-specific indications.
In this blog, I would like to discuss about two molecules many gullible veterinarians have been using as phosphorous tonic in their practice because of the marketing claims. These are: 4-dimethylamino-2-methyl-phenylphosphinic acid [Toldimfos], (available in the market as Tonophsphan, Injectiphos, Tonoricin, Tonogil, Lyphos, phosfovet, Rumiphos, T-Phos, Tenophos, etc.) and 1-butylamino-1-methyl-ethyl -phosphonic acid [Butaphosphan] (available under trade names, Cynophan, Catosal, etc.). From the list it will be clear that there are leading brands by multinational and clones marketed by local companies. The indicated claims in their brochure and literature are: phosphorous deficiency, hyophsophatemia, rickets, Ca-P imbalance, debility, anoestrus, and several other non-specific indications.
In early 2000, my research interest was to elucidate mechanism of haemolysis in phosphorous deficiency haemoglobinuria in buffaloes, especially the role of inorganic phosphorous in synthesis of ATP in erythrocytes. The team of brilliant post-graduate students developed an in vitro model of the disease. We developed a novel and easy method to remove phosphorous from the plasma, without disturbing other constituents. When the erythrocytes were incubated in this plasma for 24-36 hours, we could simulate lesions such as, Heinz bodies, depleted ATP and 2-3 DGP. The model also enabled us to track the destination of inorganic phosphorous in the erythrocytes by using radioactive phosphorous.
The next step was to validate therapeutic claims of various phosphorous-containing formulations. The evaluation criteria were to check if the co-incubated erythrocytes replenished phosphorous and if other cellular and biochemical lesions were reversed. To our surprise, although various combinations of inorganic phosphorous formulations showed excellent results, toldimphos was found to be totally ineffective.
Search of the molecules in pharmacopeia
I then started searching the details of the parent molecule, although it was not an easy task as Google by then was not available. I contacted the company through one of the senior executives. They however were sceptical and gave evasive replies, reiterating that the drug was in use for so many years, the veterinarians were very happy with the product and that the market worth of the product was several hundred (may be thousand, I don’t know) crores, per year. The point emphasized was ‘international brand’ and high-quality standards. They even refused to share the literature on pharmacokinetics and other properties. I also asked them a simple logical question to explain how a formulation containing only 200 mg phosphorous in its dose could replenish 15 g deficient phosphorous in the animal body. Instead of replies. in no minced words the needle of suspicion was on the research team.
Through a contact in Europe, I could get a page copy of an East European country’s pharmacopeia on this molecule. I was stunned to find it to be an organic ‘phosphinate’. It has a P=O bond which is not known to be hydrolysed by any mammalian enzyme. So, metabolically, I was certain that, this compound could not be a source of phosphorous. As per the data, the drug was reported to follow one-compartment kinetic, as after 6 hours, 100% of the drug was found to be eliminated in urine, that too ‘UNALTERED’. It known to be not even distributed to any organ or tissues. It then became clear why we were not seeing any effect in our in vitro system. As usual, these observations were recorded in the student’s thesis and forgotten. In my extension lectures, however, I kept on informing the veterinarians about ineffectiveness of these formulations, but I guess, in comparison to company’s marketing energy, academicians can’t hold their feet. The other molecule ‘butaphosphan’ is also a phopshinate, chemically and metabolically similar to toldimphos, but the companies marketing this product also claim similar indications, quite a few of those are unfounded with no quality data forthcoming.
Additional confirmation
While searching on the internet I accessed a report published by the European Agency for Evaluation of Medicine for Veterinary Use on these molecules (Summary Report of Committee for Veterinary Medical Products). The report made two important observations about these molecules. (1) The precise mechanism of action is unknown and is questionable- (especially the phosphorus substitution) and (2) 99% of the injected compound could be recovered in an unaltered form in the urine).
I felt vindicated and very happy when I recently read about this drug recently in the MSD Veterinary Manual, and then decided to write to write this blog. I quote the text therein;
‘In cattle, solutions containing not phosphate, but phosphinate (PO3), hyophosphite or organic phosphorous compounds such as butaphosphan or toldimphos are often used to supplement phosphorous i.v., frequently in combination with Ca, Mg and other minerals. However, these phosphorous compounds are not suitable to correct hypophosphatemia, because mammals are unable to convert phosphite or other above-mentioned organic compounds into phosphate (PO4) and so do not contribute to the biologically active plasma pool. Even when organic phosphate compounds are usable metabolized, pharmaceutical product containing organic phosphate compound administered at the label dose does not provide enough phosphorous to cover serum phosphorus depletion, which would be a primary indication for i.v. treatment’.
MSD, a market leader of this molecule (Tonophosphan), deserves compliments and kudos for their professional courage in publishing these facts very explicitly. I checked the product information on the websites of all these companies but shockingly, they all continue to claim same indications, including the MSD. Apart from phosphorous deficiency, in respect of other indications, there appear to be compelling grounds to raise serious questions and objections. The European Drug Evaluation Agency in its reports also mentioned that up to 1920’s the drug was used in humans for treating debility, weakness and post-surgery, but eased out due to the poor- quality data on efficacy and difficulty in interpretations.
In the light of all these facts, these companies owe responsibility to put before the profession the clinical data to explain how these molecules are effective in rickets, nutrition-associated -debility, post-parturient complications, etc. There are reports that after administration of the drug there is temporary elevation of blood glucose probably because the molecule is highly charged and is likely to reversibly bind the cellular receptors to stimulate glycogen hydrolysis.
In conclusion, I leave it to the wisdom of the veterinarians; facts are laid before them to take informed decisions. Can we now hope that the companies will actively and with equal zeal re-inform the veterinarians and uninstall misfeeding carried out over last so many decades?
